In the human body, there are many various systems. Each system plays a specific role in maintaining the activities of our body. Sometimes, your friends describe several different systems, which are very important. However, you have no idea about chemical buffering systems.
Don’t worry. This article will provide you with detailed information on the human body’s chemical buffering systems. Which of the following is not one of the body’s chemical buffering systems? Let’s find it out right now.

The Body’s Chemical Buffering System Definition
Before answering the core question in this article, we will go on with the definition of the body’s chemical buffering system.
Proper biological function is essential to an overall healthy body. This can be achieved by maintaining a neutral pH balance, generally around 7.4. If the pH were to drop below 7.4, it would be considered acidotic, and in case it were to rise above 7.4, it would be considered alkalotic.
The body regulates this in many ways by creating buffering systems that ultimately maintain homeostasis throughout the body’s various types of fluids and tissues.
A buffer can be thought of as similar to the mopping of a soaked cleaning cloth. Buffers are created with specific makeup and are designed to absorb pH changes that often happen in the human body when there are too many hydrogen ions or hydroxide ions.
Thus, such buffering systems can be considered the mop for hydrogen ions. They absorb them, so they don’t cause problems and protect important areas within the body, specifically for various organs like the brain and heart.
The different buffer systems in your body have all high efficiency but their operation rates are different. Changes in pH can happen within seconds in some areas to correct any imbalances in either direction. For example, the lungs will adjust their pH levels by exhaling carbon dioxide from the body. Alternatively, the kidneys may excrete hydrogen ions in exchange for sodium ions and retain bicarbonate ions to adjust blood pH.
So to speed up this process, our body can use many chemical buffering systems. These would include sodium chloride – better known as table salt, bicarbonate ions, and carbonic acid, which collectively raise the pH level of our blood to a healthy value of around 7.4.
Which of The Following Is Not One Of The Body’s Chemical Buffering Systems?
Our body has three main internal mechanisms that help us maintain the pH of our bodily fluids. The carbonic acid bicarbonate buffer system, the phosphate buffer system, and the protein buffer system work together to keep our bodies stable.
Therefore, if you hear other systems that are not three above, they will not be the body’s chemical buffering systems. This article will show you a description of three chemical buffering systems in detail.
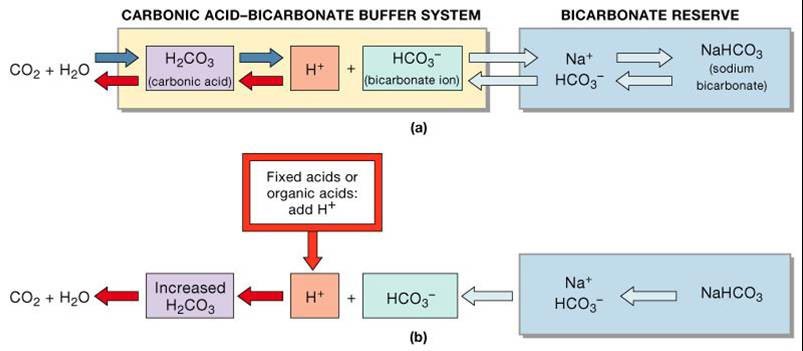
Carbonic Acid Bicarbonate Buffer
To maintain the pH homeostasis of the human body, it can lack the participation of the carbonic acid-bicarbonate buffering system. This system comprises sodium bicarbonate, which can act either as a weak acid or a weak base. Carbonic acid (H2CO3) is an anhydride that releases two protons and two bicarbonate ions when dissolved in water.
While normal respiration releases carbon dioxide into the blood, which travels throughout the body and is exhaled as waste, cellular respiration also produces carbon dioxide.
The problem is that, during cellular respiration, rather than simply releasing this gas into the atmosphere, it’s stored within the cell to keep all of the important compounds inside at a neutral pH. That is crucial to maintaining normal bodily function and structure.
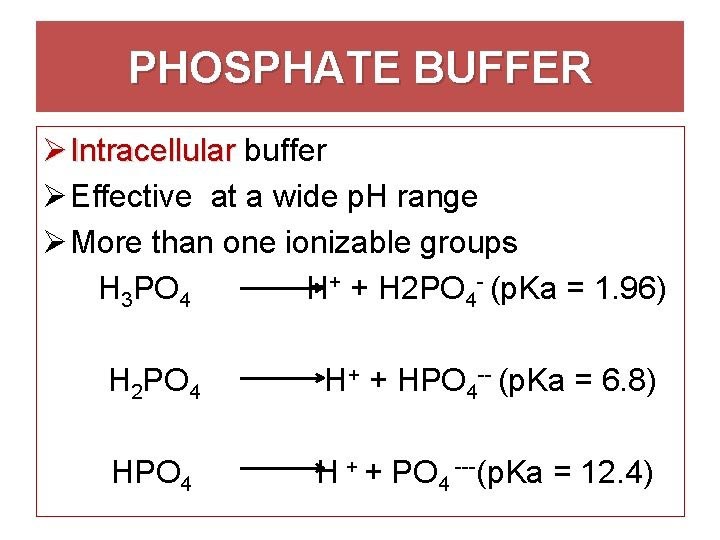
Phosphate Buffer System
The phosphate buffer system is part of intracellular pH regulation. The main purpose of this system is to keep the body’s intracellular environment stable at a pH that is optimal for cellular activity.
This system is effective because it utilizes the dual effects of phosphate, which serves as both an acid and alkaline base, to maintain homeostasis. Of interest are extracellular buffers such as hemoglobin. Hemoglobin allows for varying extracellular pH changes without the need for matching adjustments in the pH of cellular compartments.
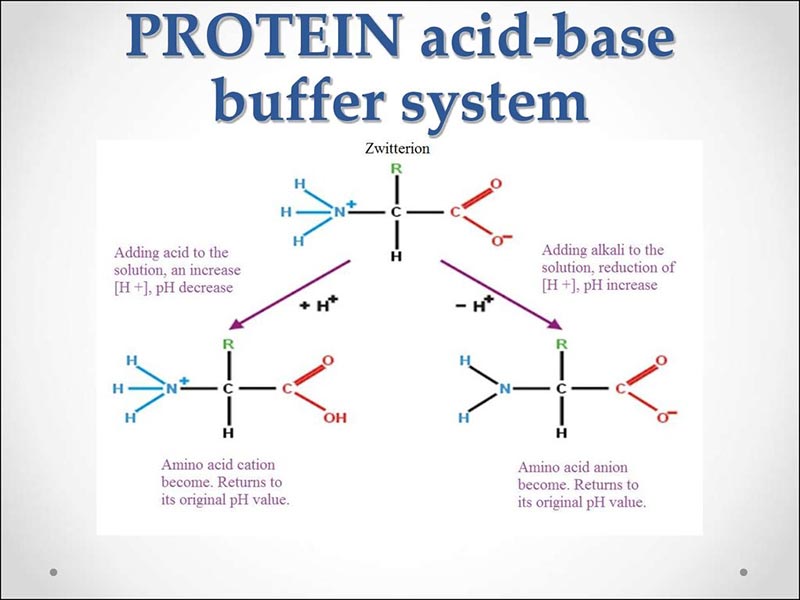
Protein Buffer System
Within the body, the protein buffer system’s role is to maintain the cells’ acidity.
Hemoglobin, the protein that makes up our red blood cells, is an excellent acid buffer. It can be observed in laboratory studies that when acidic substances are added to red blood cells, the amount of base needed to neutralize them decreases because hemoglobin has already bound with excess acids in the bloodstream before they have had the chance to alter the pH value of it.
Other proteins containing histidine amino acids also act as good buffers by binding with small amounts of acids present within the bloodstream and, as such, can prevent changes in body pH levels.
Conclusion
We believe that you have already answered the question, Which of the following is not one of the body’s chemical buffering systems whenever you read articles about the human body’s systems.
Understanding buffer systems is helpful for you to take care of your health. We hope that this article has given you a deeper knowledge about the body’s operation.
Last Updated on 4 years ago by George Morgan